Dangers of Diabetes Drug, Invokana
Diabetes Drug “Invokana” Linked to Lower Extremity Amputations and the Development of Ketoacidosis
If you or a loved one suffered from lower extremity amputation or Ketoacidosis after taking Invokana, Invokamet, or Invokamet XR, you may have a legal claim.
The drug Invokana has been on the market since March 2013. During that time, it has earned billions of dollars for Janssen Pharmaceuticals, and parent company Johnson & Johnson. According to the drugmaker’s website, 7 million people have been prescribed Invokana. Unfortunately, use of the drug has come at a severe cost for some patients. New studies show that taking Invokana can lead to lower extremity amputations and developing ketoacidosis, a rare, but deadly, condition that can cause a diabetic coma.
What is Ketoacidosis?
When your body is unable to produce enough insulin, your cells are unable to accept an adequate amount of glucose. Ketoacidosis is a condition in which your body begins to break down fat for fuel as an alternative to glucose.
Invokana and Invokament
In the United States, diabetes is seventh on the list of causes of death. Left untreated or unmanaged, type 2 diabetes can eventually lead to nerve damage, blindness, heart disease, kidney failure, high blood pressure, and other serious problems. Drugs such as Invokana and Invokamet help the kidneys remove excess blood sugar through the urine.
These drugs are SGLT2 inhibitors. When they are combined with a prescribed diet and exercise regimen, the drugs help the patient control their diabetes by lowering blood sugar and improving glycemic control. Invokamet differs slightly in that it is a combination of Invokana and metformin. It is utilized when either drug is not sufficient in controlling diabetes on its own.
2015 FDA Warning Regarding Invokana
On May 15, 2015 the Food and Drug Administration (FDA) issued a safety warning that type 2 diabetes drugs, like Invokana and other SGLT2 inhibitors, may lead to ketoacidosis.
2017 F.D.A. “Black Box” Warning Regarding Invokana
In May of 2017, the FDA issued a public “Black Box” warning regarding Invokana. In that warning, the FDA reported that anyone taking the drug should pay close attention to any side effects they experienced.
Based on new data from two large clinical trials, the U.S. Food and Drug Administration (FDA) has concluded that the type 2 diabetes medicine canagliflozin (Invokana, Invokamet, Invokamet XR) causes an increased risk of leg and foot amputations. Patients taking canagliflozin should notify their health care professionals right away if they develop new pain or tenderness, sores or ulcers, or infections in their legs or feet.
Difficulty breathing, nausea, abdominal pain, confusion, and unusual fatigue should alert patients to seek immediate medical attention. The drug puts people at risk of developing ketoacidosis and can be fatal if left untreated.
Inovkana and the Development of Ketoacidosis
A study that was published in the April, 2015 American Journal of Emergency Medicine showed that there was a link between taking SGLT2 inhibitors (of which Invokana is a part), and the onset of ketoacidosis. This report, as well as others, were enough to convince the FDA to issue a warning.
What does Invokana cause amputations?
The trials did not answer the question of why does Invokana cause amputations. According to an article in Forbes recently, some doctors speculate that while the drug decreases blood sugar and blood pressure, it may also cause the blood to thicken and subsequently pool in the foot.
As researchers struggle to explain why the amputations occur, the FDA did make a note of which types of patients might be predisposed to amputation if they decide to take Invokana. The factors that appeared to increase the odds of amputation included a prior history of amputation, peripheral vascular disease, neuropathy and diabetic foot ulcers.
What are Side Effects of Invokana to watch for?
For those who suffer from type 2 diabetes, knowing the signs and symptoms of Invokana induced ketoacidosis may end up saving their life. As the ketones build up in the blood, pay attention to these early warning signs that something is wrong:
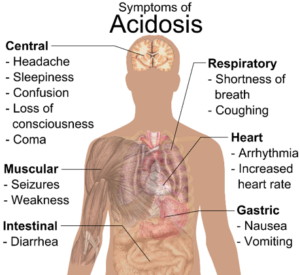
Individuals taking Invokana for diabetes may also experience these side effects:
- Bloody or cloudy urine
- Difficulty or painful urination
- Change in frequency of urination
- Bladder pain
- Increased thirst or dry mouth
- Fruity odor on breath
Although the above side effects alone are nothing to be worried about, they may be the precursors to a much more serious condition.
Below is a list of less common side effects that should also not be ignored:
- Anxiety
- Seizures
- Dizziness
- Unexplained depression
- Chills and sweats
- Hives, welts, itching skin
- Slurred speech
These worsening signs may be other signs of other issues, but there is a good chance that you are developing ketoacidosis. This condition can be stopped before it progresses too far, but steps need to be taken right away.
Filing an Invokana Lawsuit
Drug companies have a responsibility to warn their patients of all the potential side effects of a drug; no matter how rare those side effects may be. When they fail to warn the public, innocent people are harmed or even killed.
As of January 2018, more than 1,100 Invokana lawsuits have been filed against the drug maker Johnson & Johnson. More are expected.
Your Invokana lawsuit can help in two ways. First, you can be compensated for medical expenses and other damages that you may have suffered by taking a drug and not being warned about this deadly side effect. Second, your lawsuit will help to raise awareness of the dangers of this drug. Many more people may be at risk, and your lawsuit will help prevent a major health catastrophe for them.
Larry Wall Trial Law Representation
If you or a loved one has suffered an amputation or any significant health issue or complication after taking Invokana or Infokamet, contact our office at your convenience.
Each case will be handled on a contingency basis, meaning we will not accept a fee unless we settle your case in your favor. Call our offices today and tell us how we can help you.
Click here to contact us concerning your experience with Invokana.
Wichita, KS 67207
 Practice Areas
Practice Areas











